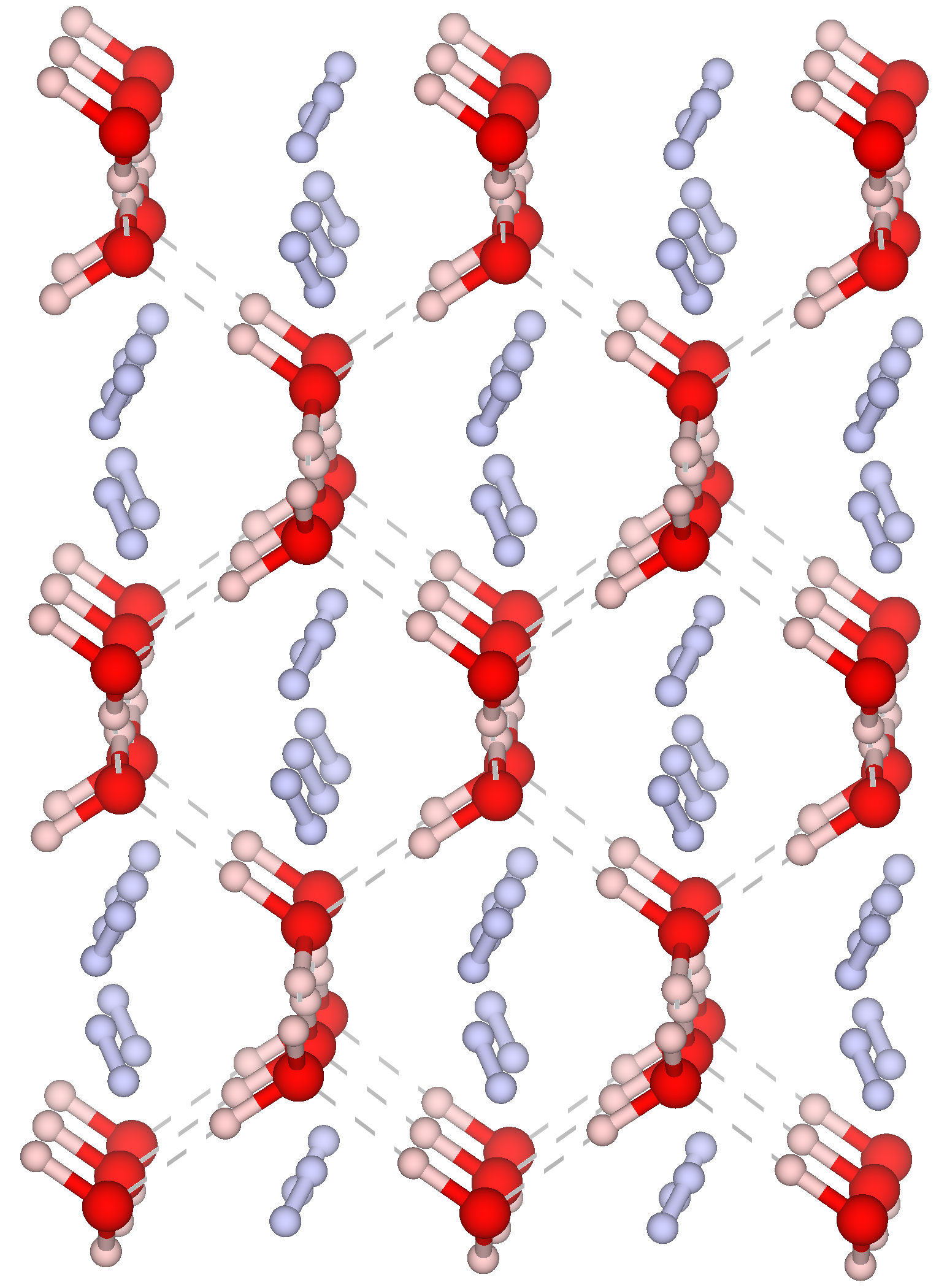
Under pressure, water and hydrogen gas spontaneously crystallize into hydrogen clathrate hydrates (above 0.1 GPa at temperatures below 270 K). These compounds are characterized by polyhedral cavities constituted of hydrogen-bonded water molecules encaging a variable amount of H2 molecules. At higher pressures, the so-called “filled ice” structures form (C0, C1, C2, C3), in which the water host sublattice is tightly wounded around the H2 molecules.
In a joint experimental and theoretical effort, the two young scientists Maria Rescigno and Simone Di Cataldo, under the supervision of prof. Livia Bove studied the fascinating rotational dynamics of H2 in these systems. This work was published in Physical Review Letters.
By combining neutron scattering studies and computational techniques, they characterized the rotational quantum dynamics of hydrogen in the C2 filled ice phase at high pressures and low temperatures (P=6 GPa, 4.5 K < T < 85 K). This compound represents one of the most extreme cases of confinement for hydrogen molecules, and thus provides an ideal benchmark to study hydrogen quantum motion in strong confinement.
In quantum mechanics, H₂ is a textbook example of a "free rotor," with evenly spaced rotational energy levels. Here, the molecule is subject to a strong angular field from the crystal. Therefore the 3D Schrödinger’s equation was solved for a hydrogen molecule embedded in the effective crystal field of the C2 phase. The result is a giant energy splitting of the rotational eigenenergies, indicating the strong impact of the hydrogen–-water interactions on the quantum behavior of hydrogen molecules in this extreme confinement.
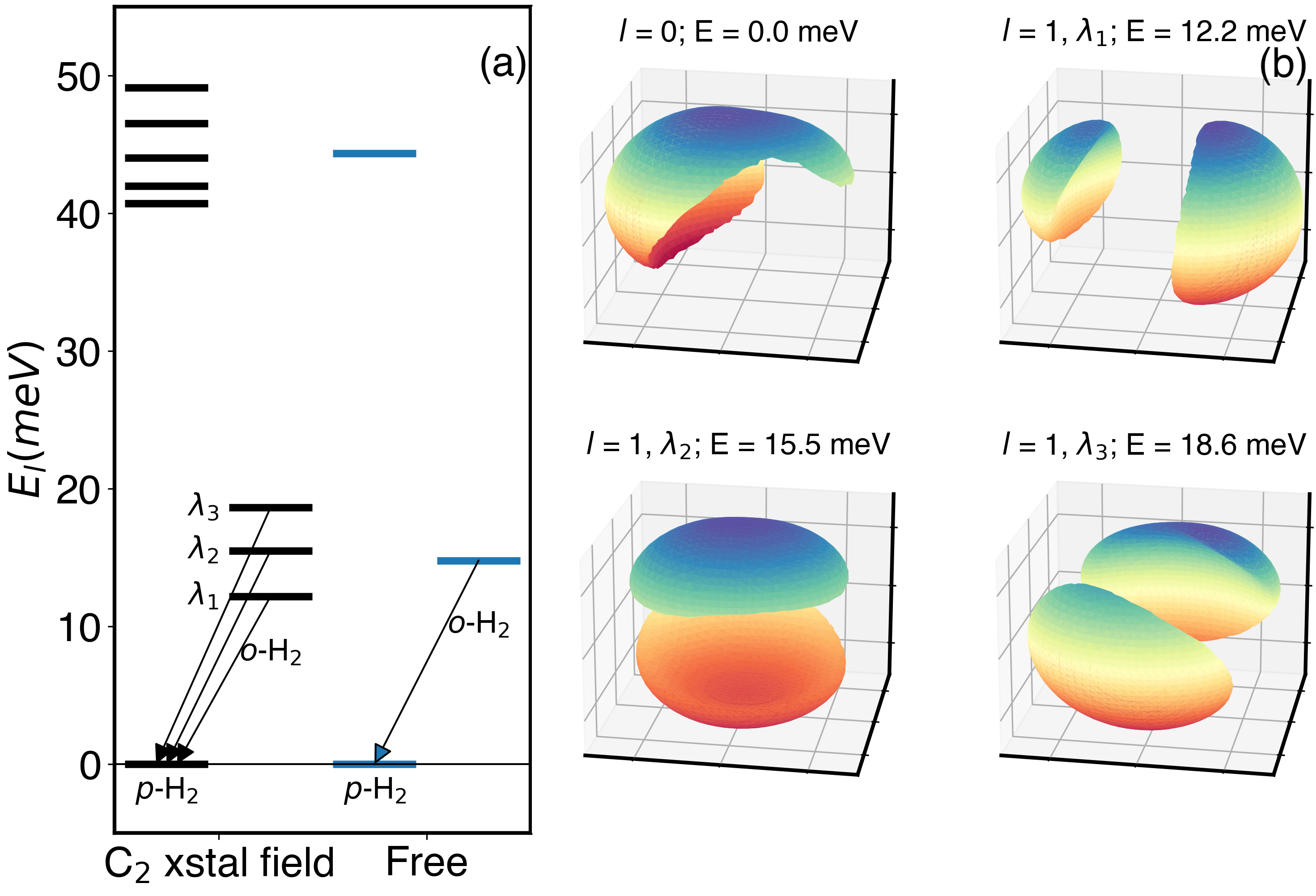
These theoretical predictions were validated through highly challenging inelastic neutron scattering experiments performed at the Institute Laue-Langevin (ILL) under extreme high-pressure and low-temperature conditions, which confirmed a prediction that would have been otherwise hard to believe.
This study reveals the fascinating dynamics governing hydrogen in hydrates and sheds light on the hydrogen–-water interaction in dense systems, which has possible implications in various scientific disciplines, ranging from material science to astrochemistry.
Authors: Simone Di Cataldo, Maria Rescigno, Lorenzo Monacelli, Umbertoluca Ranieri, Richard Gaal, Stefan Klotz, Jacques Ollivier, Michael Marek Koza, Cristiano De Michele, Livia Eleonora Bove
Magazine: Physical Review Letters
