Water’s unique thermodynamic anomalies, manifesting even under Earth's ambient conditions, hold the key to understanding fundamental mechanisms driving life and Earth’s processes.
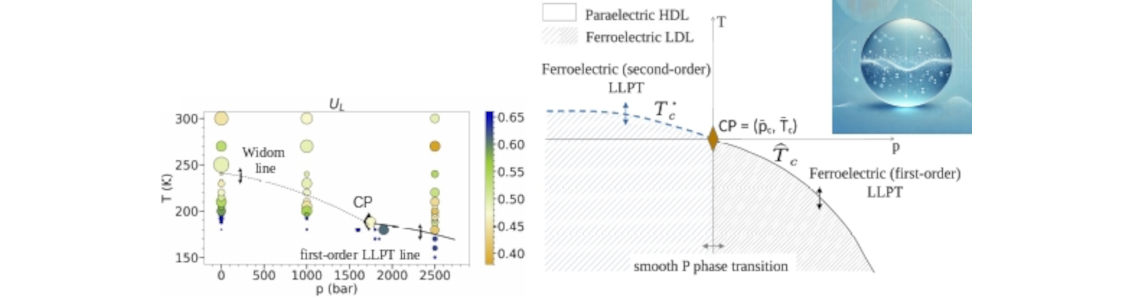
A compelling hypothesis attributes water's equilibrium anomalies to a liquid-liquid phase transition (LLPT) between high-density and low-density liquid (HDL/LDL) phases in its supercooled, metastable state. Several molecular dynamics simulation studies provided evidence for it. Nevertheless, its origin has been so-far not clarified. On the other hand, water is a polar liquid and, as such, can, in principle, undergo a ferroelectric phase transition.
Through analyzing extensive molecular dynamics simulations and developing a classical density functional theory in mean-field approximation, we unveil not only a link between ferroelectric and liquid-liquid phase transitions, but also the role of ferroelectricity in steering the liquid-liquid phase transition. Grounded in the characteristics of the microscopic dipolar potential interaction, which underpins density-polarization coupling, and liquid’s molecules positional disorder, our theory leads to a free energy expression designing ferroelectric and liquid-liquid phase transitions as two facets of the same underlying phenomenon. This not only characterize but shed light on the origin of the LLPT and the thermodynamic anomalies in water.
By demonstrating that the overlooked dipolar degrees of freedom can actually play a leading role in the liquid-liquid phase transition in water, this research seeks to open thought-provoking perspectives.
Authors: Maria Grazia Izzo, John Russo, Giorgio Pastore
Magazine: Proceedings of the National Academy of Sciences U.S.A.